


| Transport Instructions | |||
| Collection Location | Transport Temperature | Processing Required | Timeframe |
| ED/Inpatient | Room Temperature | None | Specimen must be received by the lab within 2 hours of collection |
| Room Temperature | Allow blood to clot for 30 minutes in a vertical position. Centrifuge to separate from cells within 2 hours of collection. | Specimen must be received by the lab within 48 hours of collection | |
| Laboratory/ Outpatient/ Off-Site | Room Temperature | Allow blood to clot for 30 minutes in a vertical position. Centrifuge to separate from cells within 2 hours of collection. | Specimen must be received by the lab within 48 hours of collection |

| Ordering |
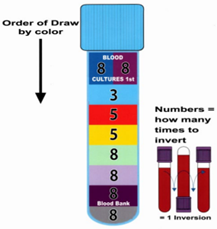
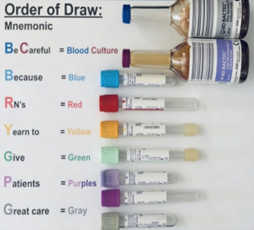
| Collection |


| Transport Instructions | |||
| Collection Location | Transport Temperature | Processing Required | Timeframe |
| ED/Inpatient | Room Temperature | None | Specimen must be received by the lab within 2 hours of collection |
| Room Temperature | Allow blood to clot for 30 minutes in a vertical position. Centrifuge to separate from cells within 2 hours of collection. | Specimen must be received by the lab within 48 hours of collection | |
| Laboratory/ Outpatient/ Off-Site | Room Temperature | Allow blood to clot for 30 minutes in a vertical position. Centrifuge to separate from cells within 2 hours of collection. | Specimen must be received by the lab within 48 hours of collection |



| Result Interpretation |